Joint Commission issues Sentinel Event Alert on inadequate hand-offs

Editor's Note The Joint Commission on September 13 announced a new Sentinel Event Alert that focuses on inadequate hand-offs and tips to improve them. A common problem with hand-offs is communication, which is the focus of “Sentinel Event Alert, Issue 58: Inadequate hand-off communication.” The alert includes an infographic of…
Effect of Medicare's Hospital Readmission Reduction Program on surgical readmissions

Editor's Note From 2008 to 2014, rates of postoperative readmissions declined for both Hospital Readmission Reduction Program targeted procedures (total hip and total knee replacements)--from 6.8% to 4.8%--and nontargeted procedures (colectomy, lung resection, abdominal aortic aneurysm repair, coronary artery bypass graft, aortic valve replacement, and mitral valve repair)--from 17.1% to…
Children’s Hospital Colorado alerts patients of data breach
Editor's Note Officials at Children’s Hospital Colorado in Aurora are sending notifications to nearly 3,400 patient families about a data breach that may have compromised their health information, according to the September 11 Becker’s Health IT & CIO Review. On July 11, investigators found an unauthorized person may have accessed…
FDA to staff new digital health unit

Editor's Note The Food and Drug Administration (FDA) is filling 13 positions in a new digital health unit that was created as part of the FDA Reauthorization Act of 2017, the September 11 FierceHealthcare reports. The FDA intends to build a group of experts who are experienced with software lifecycle…
Safety of transoral endoscopic thyroidectomy

Editor's Note The transoral endoscopic thyroidectomy vestibular approach can be safe for select patients, with outcomes similar to those of the open approach, this study finds. Of 425 patients analyzed, the operative time for the transoral approach was longer than the open approach (100.8 vs 79.4 minutes) but transoral patients…
FDA: Class I recall of Datascope/MAQUET intra-aortic balloon pumps
Editor's Note The Food and Drug Administration (FDA) on September 6 categorized the recall by Datascope/MAQUET of its CS100i, CS100, and CS300 intra-aortic balloon pumps as Class I, the most serious. The recall was initiated because of a false blood detection alarm and ingress of fluid into the balloon pump.…
Preadmission frailty predicts adverse outcomes in geriatric trauma patients

Editor's Note In this study, preadmission clinical frailty independently predicted adverse discharge destination (ie, death or discharge to a long-term, chronic or acute care facility) in geriatric trauma patients. The analysis of 266 trauma patients 65 years and older using the Clinical Frailty Scale (CFS) and the laboratory Frailty Index…
FDA: Genetech recalls three lots of Activase
Editor's Note The Food and Drug Administration (FDA) on September 7 announced the recall by Genetech (South San Francisco, California) of three lots of Activase (alteplase) 100 mg vials that were co-packaged with sterile water for injection. The vials of water, manufactured by Hospira Inc (Lake Forest, Illinois), may be…
No consensus on what defines a high-performing system
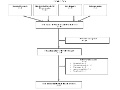
Editor's Note In this study, researchers reviewed literature from a 10-year period and found no consensus on what defines a high-performing healthcare delivery system or healthcare organization. High performance was variably defined across different dimensions, including: quality (93% of articles) cost (67%) access (35%) equity (26%) patient experience (21%) patient…
FDA: Recall of Hydromorphone HCL and Levophed Injection by Hospira
Editor's Note The Food and Drug Administration (FDA) on September 5 announced the recall by Hospira Inc (Lake Forest, Illinois) of one lot of Hydromorphone HCL injection, USP, CII 2 mg/mL and four lots of Levophed (norepinephrine bitartrate) injection because of a lack of sterility assurance resulting from a damaged…
