CMS issues guide for quality payment programs
Editor's Note The Centers for Medicare & Medicaid Services (CMS) has issued a guide summarizing technical assistance resources available to small, underserved, and rural hospitals participating in the Merit-based Incentive Payment System (MIPS) and the Advanced Alternative Payment Model (AAPM). The guide also provides resources for quality innovation networks and…
Study: QI measures reduce readmissions, cost savings vary
Editor's Note Quality improvement (QI) measures can be effective in reducing readmissions, relative to the status quo, but cost savings vary, this meta-analysis finds. In this review of 50 QI studies, interventions reduced readmissions by an average of 12.1% for heart failure patients and 6.3% among the general population. However,…
Joint Commission revises Safety Goal on HAIs

Editor's Note The Joint Commission on May 31 announced the revision of requirements for National Patient Safety Goal (NPSG) 7: Reduce the risk of healthcare-associated infections. The changes are effective January 1, 2018. The requirements include: NPSG.07.03.01−Multidrug-resistant organisms. The changes help address infection prevention strategies to contain outbreaks, which now…
Healthcare IT market projected to grow by 15.9%

Editor's Note Research N Reports predicts the global healthcare information technology (IT) market will grow by 15.9% and reach $280.25 billion by 2021, according to the May 30 Healthcare Informatics. The growth is driven by demand for healthcare IT solutions that help lower costs while meeting regulatory requirements. Other factors…
Postop program reduces delirium risk, LOS for elderly
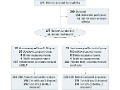
Editor's Note Older patients who had major abdominal surgery and received interventions that included daily orienting communication, oral and nutritional assistance, and early mobilization were less likely to experience delirium and had a shorter hospital length of stay (LOS), this study finds. A total of 377 patients were included in…
Joint Commission posts addendums for radiation risks of diagnostic imaging

Editor's Note The Joint Commission on May 30 posted addendums to its Sentinel Event Alert, Issue 47: Radiation risks of diagnostic imaging. Risks associated with ionizing radiation in diagnostic imaging included cancer, burns, and other injuries. The addendums update broken links, resources, and references. Sentinel Event Alert, Issue 47: Radiation…
FDA: Class I recall of Zimmer Biomet implantable spinal fusion stimulators
Editor's Note The Food and Drug Administration (FDA) on May 30 identified the recall by Zimmer Biomet (Warsaw, Indiana) of its SpF PLUS-Mini and SpF XL IIB implantable spinal fusion stimulators as Class I, the most serious. The recall was initiated because of higher than allowed levels of potential harmful…
Improving postop transitional care with phone-based program
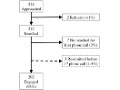
Editor's Note A phone-based transitional care program after complex abdominal surgery has high patient engagement and can be implemented using existing hospital infrastructure, this study finds. For the program, nurses met patients before hospital discharge and then contacted them within 24 to 72 hours to review: medication reconciliation any symptoms…
ECRI Institute issues free resource on ransomware attacks

Editor's Note The ECRI Institute (Plymouth Meeting, Pennsylvania) on May 25 issued a free public resource to protect hospitals from ransomware attacks. The new guidance, “Ransomware Attacks: How to Protect Your Medical Device Systems,” provides recommendations for adapting general cybersecurity principles to the requirements of medical devices, including a list…
Joint Commission: Safety update on improperly sterilized, HLD equipment

Editor's Note The Joint Commission on May 24 issued a new Quick Safety update on improperly sterilized or high-level disinfected (HLD) equipment, which continues to be a frequently scored noncompliant standard (IC 02.02.01). The update includes expanded safety actions to help leaders oversee sterilization and HLD processes and ensure that…
