
Editor's Note The US Food & Drug Administration (FDA) has announced Class 1 recalls—the most severe category, indicating risk of serious injury or death—for three products: MEGA SOFT Pediatric Patient Return Electrodes from Megadyne, Vaporizer Sevoflurane Maquet Filling from Getinge; and Arrow FiberOptix Intra-Aortic Balloon Catheter Kit and Arrow UltraFlex…

Editor's Note The US Food & Drug Administration has designated Medline Industries’ recall of the Sub-G Endotracheal Tube with Subglottic Suction, which is used to keep a patients’ airway open, as a class 1, the most severe category indicating risk of serious injury or death. According to the agency’s June…

Editor's Note The US Food & Drug Administration (FDA) has classified Medtronic’s recall of its StealthStation S8 software as a Class I, the most severe category indicating risk of serious injury or death. The StealthStation System with StealthStation Cranial software is intended as an aid for locating anatomical structures during…

Editor's Note The US Food and Drug Administration (FDA) has designated OptumHealth Care Solutions’ recall of the Nimbus II ambulatory infusion pumps a class 1, the most severe category indicating risk of serious injury or death. According to the agency’s May 30 announcement, the company recalled the pumps in direct…

Editor's Note A choking hazard prompted the US Food and Drug Administration (FDA) to designate Vyaire Medical’s recall of Twin Tube sample lines—critical components of the Vyntus CPX system—as Class 1, the most serious category of recall indicating a risk of serious injury or death. According to the agency’s May…
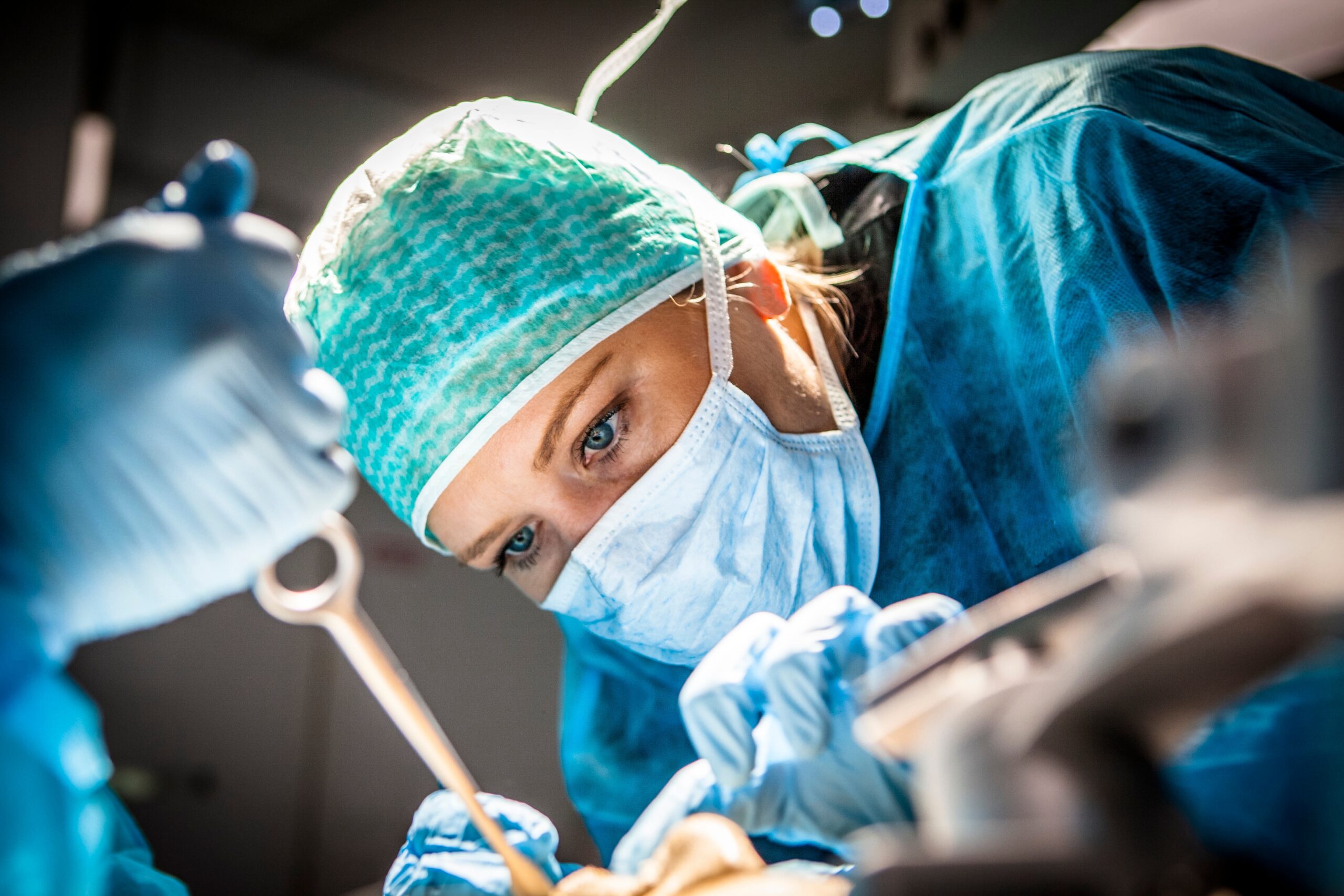
Takeaways Although women comprise half the population, they were left out of medical research on major causes of death for both women and men—cancer, heart disease, and stroke—until 1990. Using surgical tools designed by men, for men can impact every aspect of a woman surgeon’s work, from learning new procedures…

Editor's Note A recent trial marks the last hurdle for researchers to request regulatory approval for a noninvasive alternative to spinal surgery, according to a May 20 report in MIT Technology Review. Onward Medical’s ACRex device delivers electrical stimulation to the spinal cord via two wired electrodes placed just below…

Editor's Note The largest scale analysis so far available comparing surgical aortic valve replacement (SAVR) to longer-term percutaneous devices for transcatheter aortic valve replacement (TAVR) supports the comparable long-term safety and efficacy of the latter procedure. According to a May 15 report in Medical Xpress, the findings raise important considerations for valve…

Editor's Note More than 45 years after surgery, Mary Ann Kozlowski’s mechanical heart valves are still pumping—and Guinness is reviewing an application to list her as the new world record-holder for longevity of a double-valve replacement. That’s according to a May 15 report in the Erie-Times News, which detailed how…

Editor's Note The US Food and Drug Administration (FDA) has classified recalls of Abbot’s HeartMate 3 Left Ventricular Assist System (LVAS) implant kits and Phillips Respironics' Trilogy Evo ventilators as class 1, the most serious category indicating risk of serious injury or death. Used while waiting for recovery, a transplant,…